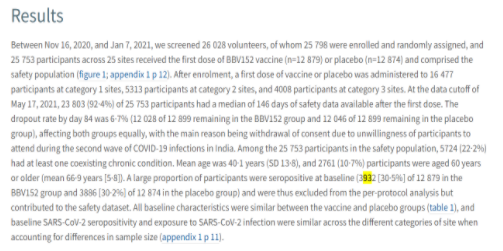
Immediately after receiving a much-delayed Emergency Use authorization (EUA) approval from World Health Organisation (WHO) medical journal Lancet has now stated that the made-in-India vaccine is 77.8 per cent effective against symptomatic Covid, as demonstrated by the phase 3 data. The efficacy data demonstrated 70.8 per cent protection against all variants of the SARS-CoV-2 virus. More importantly, the vaccine showed 93.4 per cent efficacy in severe symptomatic cases.
The medical journal added that no severe-vaccine-related deaths or adverse events were recorded during the randomized trial. The phase 3 trial efficacy and safety study involved 25,800 volunteers across 25 sites between November 2020 and May 2021 in India and is the country’s largest-ever clinical trial conducted for a Covid-19 vaccine.
Elating the results, Bharat Biotech chairman Dr Krishna Ella said, “The peer-review of Covaxin phase III clinical trial data in The Lancet, an authoritative voice in global medicine, validates our commitment to data transparency and meeting the stringent peer-review standards of world-leading medical journals. This accomplishment reflects the undeterred commitment by my team members at Bharat Biotech, our public partners, Indian Council of Medical Research, National Institute of Virology, and the trust imposed by our trial participants who made this happen.”
Negative Nancy’s had delayed the process
Covaxin has cleared all the hurdles despite facing stiff resistance from a massive foreign pharma lobby, a dud opposition that plays politics even in a life-or-death situation and a bought-out media, that leaves no opportunity to target the government through hook.
As reported by TFI, a couple of days ago, Dr Ella whilst speaking at Times Now Summit had remarked that it was due to the negativists in the country who had called Covaxin a BJP vaccine amongst other cheap names that led to the delay in the approval process.
Ella said, “They (WHO) wanted to be doubly sure of what they were doing. So, they wanted to review much more intensely. Every small issue became a major issue for them,”
He further added, “We are the only vaccine in the WHO that has gone through so much scrutiny which other vaccines have not gone through. But it’s good that in the end, we won the game,”
Congress and its role in creating an anti-Covaxin narrative
It was Congress leaders like Anand Sharma, Shashi Tharoor and Jairam Ramesh that raised serious hell when the government had granted emergency use of the two vaccines in India. Showing a colonial mindset, the dud Congress leaders started attacking the Indian company, Bharat Biotech, as its phase-3 results were not announced.
Senior Congress leader Jairam Ramesh had tweeted, “Bharat Biotech is a first-rate enterprise, but it is puzzling that internationally-accepted protocols relating to phase 3 trials are being modified for Covaxin. Health Minister @drharshvardhan should clarify.”
Bharat Biotech is a first-rate enterprise, but it is puzzling that internationally-accepted protocols relating to phase 3 trials are being modified for Covaxin. Health Minister @drharshvardhan should clarify. pic.twitter.com/5HAWZtmW9s
— Jairam Ramesh (@Jairam_Ramesh) January 3, 2021
Similarly, thesaurus Shashi Tharoor tweeted, “India actually has a good reputation in the vaccine world, both as a producer(60% of the world’s vaccines) &as a consumer (the success of the polio & smallpox vaccination campaigns). This unseemly haste risks jeopardising both, especially if Covaxin turns out to be ineffective.”
The negative Nancy in Shashi Tharoor came out in full glory when he remarked, “Worse, if it turns out to be unsafe. But it is typical of a govt that prefers slogans over substance. Chest-thumping ‘vaccine nationalism’, combined with the PM’s “atmanirbhar Bharat” campaign, has trumped common sense and a generation of established scientific protocols.”
. Third: "We are baffled to understand what scientific logic has motivated the SEC to approve this vaccine posthaste… in violation of the criteria in the draft regulatory guidelines for development of Covid-19 vaccines published by CDSCO on 21/9/20." https://t.co/XVi7Yvf6R8
— Shashi Tharoor (@ShashiTharoor) January 3, 2021
Srivatsa, another Congress worker had called Covaxin a jumla vaccine. The likes of Tharoor, Jairam Ramesh and other Congres leaders were not concerned with the health of the public, no. All they wanted was to besmirch the name of the government by making a false accusation and innocently planting the idea in public that the vaccine was another PR stunt by the NDA government.
Pfizer, Moderna & Oxford Vaccines
✅ Phase 3 Large Scale Efficacy Tests
✅ Test Data was Peer Reviewed
✅ Being used by many countriesBharat Biotech's Covaxin
❌ Phase 3 Efficacy Tests not done
❌ Data not shared for peer review
❌ Not being used anywhere elseJumla Vaccine?
— Srivatsa (@srivatsayb) January 3, 2021
However, ever since the test results by the Lancet have been published, the entire opposition has preferred to observe a stoic silence.
2 BHK Pfizer and left-media lobby
Meanwhile, enrolled on payrolls of the Pfizer lobby and masquerading as a journalist, Rohini Sharma had opened an entire front to bat for the US-developed overpriced vaccine. In one of her tweets she had questioned, “Why hasn’t the Indian government not given permission to Pfizer vaccine yet?”. Meanwhile, in several other tweets, Rohini remarked that she would have preferred taking the Pfizer jabs.
https://twitter.com/MVAGovt/status/1455874645027033094?s=20
It wasn’t the opposition leaders only that tried to disparage the vaccine. As reported by TFI, it was NDTV and its Harvard alumni journalists were also leading the charge of creating an environment of psychosis when it dedicated a full section on its airtime to highlight the one or two odd cases that prop up whenever someone took a vaccine.
And even in one of the cases where the reporter talked to a patient, he accepted he had forgotten to take his blood pressure medicine in the morning, before taking the vaccine.
NDTV has posted video with title
"Corona Vaccine लगवाने के बाद स्वास्थ्य कर्मी की बिगड़ी तबीयत"
In the video, the man says (in Hindi) : I had forgotten to take my blood pressure medicine, so I felt uneasy and got admitted. Everyone should take the vaccine.
Cheer up, NDTV!
— Abhishek (@AbhishBanerj) January 17, 2021
Meanwhile, the Print, often monkey-balancing between its allegiances had initiated a vicious campaign to attack Covaxin. In myriad op-eds, Shekhar Gupta and his team conducted hitjobs on the Indian vaccine.
From headlines like, “DGCI’s Covaxin approval is political jumla. It reinforces idea of Modi’s Aatmanirbhar bharat” to “This is how Bharat Biotech changed expert panel’s mind on Covaxin in a day and got approval” — the Print left no opportunity unturned to punch down on Bharat Biotech.
Covaxin found to neutralise 617 variant of Covid-19: Dr Fauci https://t.co/iKggG3QSIF#NeverForget and #NeverForgive @ShekharGupta for his nefarious, criminal and motivated campaign against Covaxin via his platform @ThePrintIndia / here are some samples from Jan 21 pic.twitter.com/mmXTN4WJ44
— Alok Bhatt (Modi Ka Parivar) (@alok_bhatt) April 28, 2021
India Today journalist Shiv Aroor took a swipe at lobby members of the big pharma companies after the announcement of phase three results and remarked, “If you listen carefully, you can hear pharma company dalals guised as analysts rolling up their car windows”
If you listen carefully, you can here pharma company dalals guised as analysts rolling up their car windows https://t.co/RQLvmtj9z6
— Shiv Aroor (@ShivAroor) November 12, 2021
While the opposition and media tried their best to create an anti-vaccine environment, Bharat biotech and the government kept their head down and steadily rolled out the jabs. After the success of Covaxin in every single trial and nearly 100 countries allowing it, the pharma lobby must be sweating hard at the prospect of facing a much efficient and much popular vaccine in the competition.